FDA: Plan for Oversight with ML/AI in Medical Devices
CONTEXT: FDA publishing an action plan on AI in SAAMDs. China already ahead of the game in terms of how they have already enacted guidance on AI in SAAMDs, but Continue Reading

CONTEXT: FDA publishing an action plan on AI in SAAMDs. China already ahead of the game in terms of how they have already enacted guidance on AI in SAAMDs, but Continue Reading
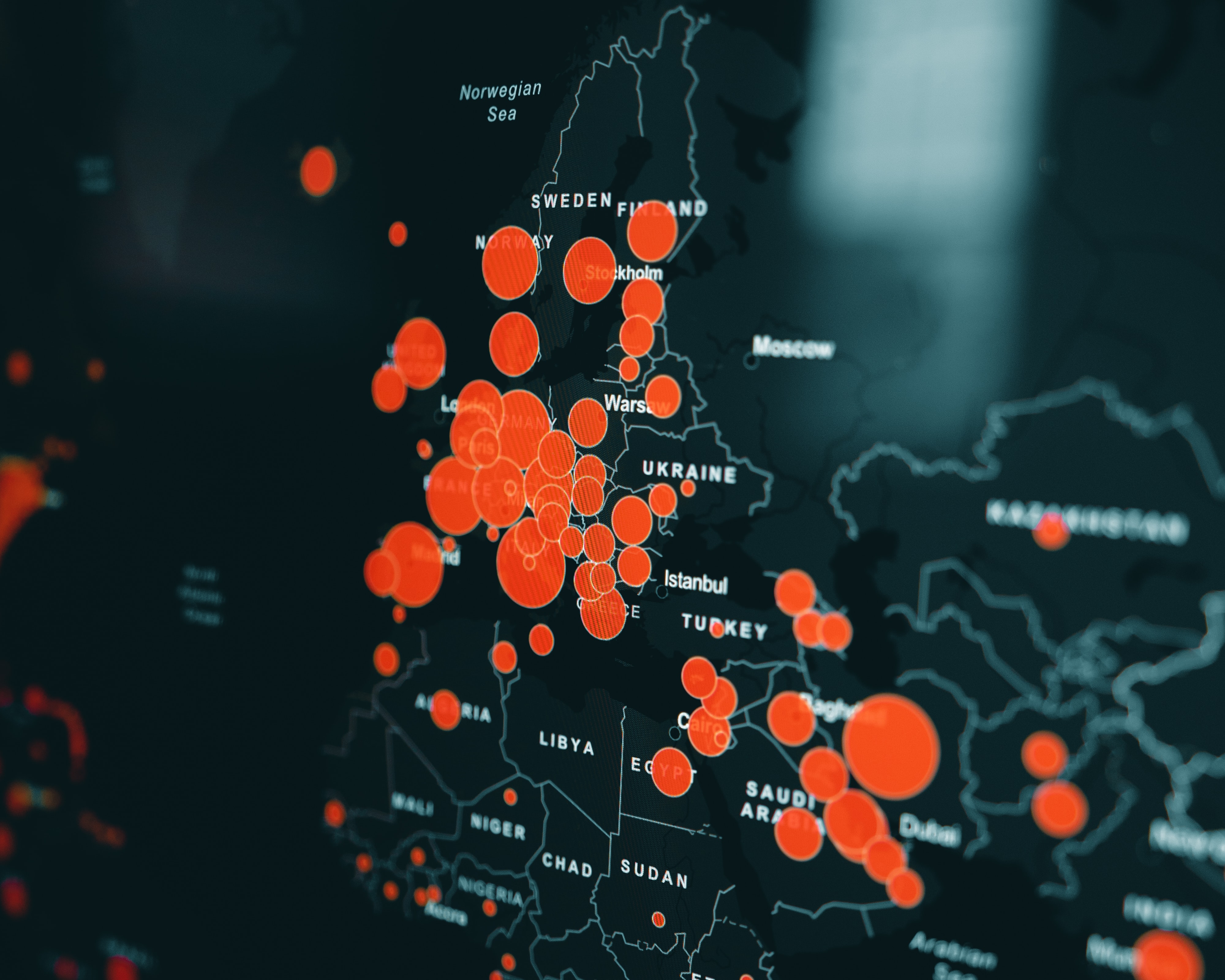
Comments – Part of the EMA’s continued commitment to RWE in support of medicinal product utilisation. When you see regulators investing in infrastructure like this, you know they are serious Continue Reading
COMMENTS: Part of the growing trend for big pharma to secure bid data sources for their own use. IMPACT: A move in the right direction towards ‘regulatory grade’ RWE, READ Continue Reading

IMPACT: Taking insulin and a GLP-1 agonist significantly increases the ability of achievement stable blood sugar levels READ TIME: 2 mins * 1. “Sanofi’s Soliqua combines an insulin with a Continue Reading

Quick Notes: CONTEXT: RWD, RWE, Pfizer, Isaeli Government, Data Transparency, Real-World Epidemiological Evidence Collaboration Agreement IMPACT: Sharing of Real-World Epidemiological Evidence to improve understanding of impact of COVID-19 vaccination Time Continue Reading

Quick Notes: CONTEXT: USA, IVDs, RWE, Real-World Evidence Framework for regulatory decision-making for in vitro diagnostics (IVDs) IMPACT: step forward in providing the valuable information that IVD manufacturers and others Continue Reading

EXPERT NOTES: CONTEXT: EU, Commercial RWD Data Source IMPACT: Access to EU RWD, drive cutting edge improvements in patient outcome Time Taken = 3 mins QUOTES FROM SOURCE: 1. “NEW Continue Reading

EXPERT NOTES: CONTEXT: Peer Reviewed Data, RWE of Effectiveness of Cabozantinib IMPACT: In the largest real-world study to date, cabozantinib was effective in unselected, heavily pretreated patients with mRCC. Initiation Continue Reading

EXPERT NOTES: CONTEXT: Decentralized Clinical Trials IMPACT: Enabling technology for ‘Decentralized Clinical Trials’ a.k.a ‘Virtual Trials’ Time Taken = 2 mins QUOTES FROM SOURCE: 1. “As mentioned earlier, COVID-19 has Continue Reading

Quick Notes: CONTEXT: Japan, Real-World Data IMPACT: Acceptability of RWD in Regulatory Submissions Time to Read = 2 mins Quotes from Source: 1. “TOKYO — Japan’s health ministry will issue Continue Reading